It is only when the electrons are flowing from the negative end to the positive terminal that the chemical reactions occur. This is the main reason why batteries are able to sit a shelf for long periods of time and still retain their energy source. The electrons start to flow into the wire, after which they will travel from the negative end to the positive terminal to initiate the chemical reactions. The rate of electron production during the chemical reaction determines how much electrons are able to flow through the terminals. Within the internal structure of a battery, the chemical reactions will produce electrons. A load can be a light bulb, a motor or even an electronic circuit. Generally you will have to connect a load to the battery while using the wire. This process will deplete the energy levels of a battery rapidly and as the battery size increases, the energy-creating process becomes more dangerous. Whenever a wire is connected between the negative & positive segment of a battery, the electrons will start to flow towards the positive terminal as quickly as possible. Usually, polarization of electrons is found on the negative terminal segment of the battery.
This is something that many investors are eying as the look to the future of lithium. The demand of lithium will also see a spike in the coming years as more and more lithium is needed to meet that demand. Green cars are slowly taking over diesel cars as the world becomes more informed about global warming and other environmental concerns. In fact, even the electric car is powered by lithium. It can be found within the battery of your Mp3 or within the circuitry of your laptop. There are hundreds of ways that lithium has become integrated into our lives. Lithium fluoride (highly enriched in the common isotope lithium-7) forms the basic constituent of the preferred fluoride salt mixture (LiF-BeF2) used in liquid-fluoride nuclear reactors. Lithium deuteride was the fusion fuel of choice in early versions of the hydrogen bomb. Its complex hydrides, such a Li, are used as high energy additives to rocket propellants.

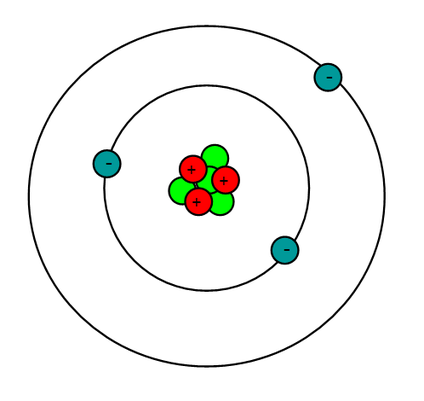
Sometimes used in focal lenses, including spectacles. Lithium fluoride, artificially grown as crystal, is clear and transparent and often used in specialist optics for IR, UV and VUV (vacuum UV) applications. Promotes the fusing of metals while eliminating the forming of oxides by absorbing impurities from the base elements. Lithium stearate is a common all-purpose, high-temperature lubricant. Used in preparation of organo-lithium compounds to create strong bases in opposition of strong alkaline composites. Lithium chloride & lithium bromide are extremely hygroscopic (the ability of a substance to attract water molecules from the surrounding environment through either absorption or adsorption.) and are used as desiccant (Induces or sustains a state of dryness in its local vicinity in a moderately well-sealed container). Helium has an atomic number 2, Neon has an atomic number of 10, while Argon has an atomic number of 18.

So, in the first 20 elements, there are three noble gases, namely, Helium (He), Neon (Ne), and Argon (Ar). The noble gases were characterized later than other elements. Lithium niobates (lithium, niobium and oxygen) are used extensively in telecommunication products such as mobile phones and optical modulators. These are known to be the least reactive or extremely non-reactive.
Lithium batteries are usually disposable with lithium compounds as an anode. 2009.Lithium is Used For: Electrical and Electronics "periodic table of the elements." Praire hill middle school student handbook 2009-2010 Aug."Chemical - Lithium (Li)." Chemical - An Interactive Periodic Table of the Elements.The Physical properties of Lithium is to the left. Interesting Facts: Lithium is the lightest known metal ( like a feather) It can make batteries it also can reacts water and sodium it also releases Hydrogen gas.
